Air conditioning
Clinical requirement underpinning recommendations
Based on current advice from the Clinical Excellence Commission, special environmental controls, such as negative pressure isolation rooms or negative pressure air flows within an entire clinical unit, are not necessary to prevent the transmission of COVID-19 as the pathogen is spread via contact and droplet. The need for negative pressure controls are recommended for aerosol generating procedures but where not available alternative of single room with door closed is sufficient. It is important to note the aerosolisation of droplets does not create airborne spread. This advice is consistent with National and International recommendations.
The use of standard bed spaces
The overwhelming majority of patients with COVID-19 can be managed in standard clinical environments without any change needed to air handling systems.
Health services should check existing air handling systems to:
- Assess the existing configuration, air flows, air changes and pressures
- Ensure the air conditioning and ventilation system is functioning properly
- Replace existing filters with new for this purpose. Monitor filter pressure drops across filters at weekly intervals to ensure that filters are functioning and replace filters when the pressure drop reaches 80% of maximum allowable limit.
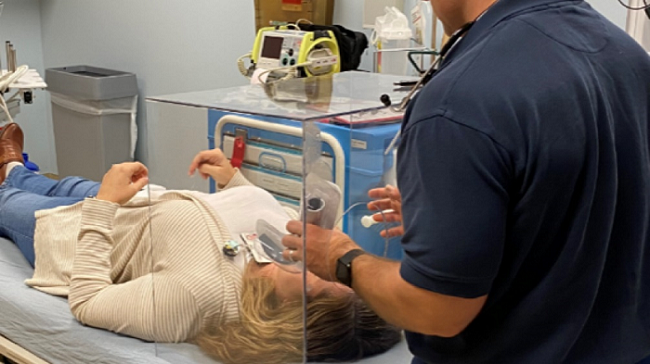
Negative pressure rooms can be used to accommodate patient care requiring AGP. An alternative is the use of a hood as shown in Figure 2.
Figure 2: Hood for AGP
Negative pressure environments
On the basis that there is a need to increase the number of rooms/areas with a negative pressure environment to support the expected increase patient numbers, the following advice should be considered.
Where a room is built with its own return/ exhaust, adjustments could be made easily, and without significant cost, to generate negative pressure. The majority of hospitals have operating theatres with their own return/ exhaust. All rooms should have a dedicated air handling system, the existing arrangement would have a supply air volume in excess of the return/ exhaust volume to generate a positive environment for the normal functioning of the operating theatre.
If both supply and return/ exhaust air volumes were adjusted and rebalanced by adjusting the fan speeds/ balancing dampers, the positive pressure can be reversed to a negative pressure environment. This will not be equivalent to a Class N isolation room but will prevent air from this room from flowing outwards. To provide visual indication of pressure differential, a wall mounted differential gauge can be installed on the outside wall of each room that is converted for this purpose (e.g. magna helix guage).
Hospitals with air handling systems serving multiple theatres, should be excluded from this use.
All existing operating theatre systems have terminal HEPA filters in the supply air outlets in the theatre (some older systems may have in-line filters) and these can remain in operation.
Check all exhaust discharge points are in correct and compliant locations in accordance with relevant standards.
While this approach is recommended for single rooms or spaces, should the approach be considered for larger areas which are equipped with dedicated exhaust fan and discharge path, then advice should be obtained by a mechanical engineer, Health Infrastructure can provide recommendations - contact details are in the introduction.
Refer to the following section for additional considerations relating to ventilation.
Other engineering and related considerations
| Oxygen |
All clinical bed spaces are equipped with at least one oxygen outlet.
Under the expected high demand of every patient being intubated, the local distribution pipework may not support volumes and pressure, resulting in low pressure/flow at the outlets, as the pipework have been designed for normal diversified operation. Test flows in pre commissioning, and as a contingency back-up, additional portable oxygen bottles can be used for a portion of the beds to reduce the load on the distribution pipework and enable all beds to have proper flow.
If oxygen bottles were to be deployed, then a storage area will need to be established, with clear demarcation between full and empty bottles. Its location should facilitate easy delivery of new bottles and removal of empty bottles.
All hospitals of a certain size and scale will be equipped with centralised oxygen supply, the overall consumption may increase thus will result in more frequent delivery and refilling. Most tanks have remote monitoring and automatic ordering system with the suppliers. Check and ensure that this is functioning properly.
Consideration should be given to undertaking a site medical gas performance capability test to determine the maximum infrastructure capacity available. This test may assist to inform the planning and use of supplementary bottle supply and use strategies. This test however should not delay the progress or completion of works.
Ensure compliance with Australian Standards AS2896-2011 and NSW Health PD_2017_007 Managing the installation, refurbishment and commissioning of medical gas systems
|
| Suction |
All bed spaces are equipped with at least one suction outlet.
• Under the expected high demand for ventilation, both the local distribution pipework and the central suction vacuum pumps may struggle to keep up, resulting in low pressure/ flow at the outlets, as the systems have been designed for normal diversified operation. If this was to occur, individual local suction pumps can be used for a number of the beds to reduce the load on the system and enable all beds to have proper flow.
|
Medical air
|
All beds are equipped with at least one medical air outlet. It is expected the system will be able to cope and additional bottled gases will not be needed. |
| Ventilation |
All existing IPU bedrooms are provided with air conditioning with an outside air component complying with regulatory requirements. This is expected to be adequate for intubated patients.
With the high number of intubated patients and thus the oxygen used, the oxygen level in the rooms may/will increase. The normal atmospheric background oxygen level is 20.9%, if this was to rise to above 23.5% than it is viewed as oxygen enriched and will pose a higher ignition and fire hazard.
It is recommended that portable oxygen sensors are deployed in various rooms for periods of time in each to establish the oxygen level under the desired operation regime (i.e., number of intubated patients in a room), and if necessary review the density of beds to manage the oxygen level.
|
| Electrical power supply |
Existing beds are likely to already have a high number or power outlets, some of these are supported by the emergency power system. The number and capacity are both expected to be adequate. For any monitoring or life supporting equipment, it would be recommended that the standby power supported outlets be used. No UPS is available in a typical IPU bed space and this is expected to be adequate.
|
| Body/cardiac protection |
With the exception of ICU (adult, paediatric and neonatal), cardiac theatres, cath labs and CCU beds, cardiac protection is not supplied to other bed spaces (i.e. IPU, short stay unit, recovery or general operating theatres). These spaces are equipped with body protection.
The Australian Standard AS/NZS 3003:2018 Electrical installations - Patient areas states that ‘a patient area shall only be wired as a cardiac-protected electrical area if cardiac-type procedures as determined by the responsible organization/ entity are to be undertaken on a regular and routine basis’. It is also only required in ICU bed space where ‘regular thermodilution Swann-Ganz monitoring’ also known as Pulmonary Artery catheter monitoring is conducted (p. 19).
As ICU adaptable bed spaces will not be used on a regular and routine basis, the existing body protection system will be deemed as adequate.
|
| IC&T |
Existing beds are likely to be equipped with at least one double data outlet and these are expected to be adequate. For equipment that has wireless capability and communication with a medical centralised reporting or monitoring system, these are expected to be set up already for their normal operation. |
| Fire safety |
All recent hospitals are provided with fire sprinklers, which are above code requirements to provide fire protection for patients. Fire detection and alarm systems are also provided. Consider a review of evacuation procedures if changes in layout have occurred or major change in patient acuity is planned. |
| Room temperature & humidity |
If a bed or a single bedroom was to have a higher than normal number of equipment items in operation, this may lead to a rise in localised room temperature. It is expected the air conditioning system will cope in most cases. In some cases, there may be an increase but it is not expected to become untenable. |
| Renal replacement therapy |
It is recommended that any patients that require both intubation and renal replacement therapy (RRT) be treated in ICU or other bed spaces equipped with a RO station.
|
Commissioning bed spaces
When delivering new and adapted bed spaces, the operational commissioning of these spaces should be adapted in line with standard operational policies and procedures such as (but not limited to):
- Infection prevention and control
- Work health and safety
- Environmental services including cleaning, linen and waste
- Clinical support services such as pathology
- Supply services
- Food services
- Transport (both internal and external)
- Building services (medical gases, HVAC and electrical supply)
Appendix A – Draft process to follow:
Reinstating clinical spaces that are currently not in use for patient care
For a site identified for recommissioning, the following appendix outlines the suggested process for site auditing, procurement, recommissioning.
The suggested process steps include:
1. Site/ IPU/ unit is identified as designated surge capacity work
2. Site Audit. Project team including services contractors and trades to audit site and produce return brief on
- Scope of works required to complete recommissioning in line with the brief and this guide
- Status/ condition of all infrastructure and plant/ equipment
- Procurement list
- Estimated time to completion
- Cost estimate
- Minimal changes are expected where possible
3. LHD should undertake a risk assessment regarding the extent of change. This should also consider the requirement for BCA advice. Recommendations from Health Infrastructure can be provided - contact details are in the introduction
4. At time of recommissioning, all trades to produce commissioning records for works in line with Australian Standards
- Medical gasses
- Electrical outlets and RCD testing for body certification compliance
- Communications connectivity (as applicable)
- Nurse call functionality
- Mechanical air handling systems
- Fire protection and detection systems
- Emergency evacuation operation (EWIS/ SSEIP)
- Hydrants/ hose reel systems.
5. Review building systems that support the unit to assess:
- If air handling unit is suitable, review to determine if outside air and associated relief can be increased to achieve increased air changes for excessive oxygen build up if required (where higher than normal oxygen therapy is used such as patient ventilation)
- For operating theatres that have been rebalanced to provide negative pressure environments, it should be noted that all existing settings of dampers, VSD's etc. should be locally marked and recorded to allow easier reestablishment when returned to normal usage.
6. Review of existing site asset management plan to
- Identify the associated plant and equipment to the designated surge areas to develop a temporary maintenance schedule to ensure ongoing service reliability
- Possibly include new and temporary requirements such as
i.) Intermittent monitoring of oxygen concentration levels within each bed area via handheld device
ii.) Increased maintenance frequency to supporting infrastructure.
Appendix B – Draft process to follow
Adapting clinical spaces that are currently in use for patient care
In support of the main body of this DGN, this appendix outlines the process which is recommended to be undertaken to facilitate the conversion for a space that is currently in clinical operation.
Suggested process steps include:
1. Site/ IPU/ unit is identified as designated surge capacity works.
2. Meeting scheduled with local nursing manager/ facilities manager to confirm that existing services are fully operational. If issues are discovered, these will need be investigated and repaired urgently.
3. Review of all existing bed provisions in line with Section 5 of this Guide and:
- Ensure a schedule of compliance/works to be completed and should be produced as part of the facility maintenance schedule
- Recommend the completion of a capacity test for oxygen supply capability which will provide information to inform the assessment of bottled gas requirements. This will also inform required space requirements storage issues for bottles.
4. Review building systems that support the department generally. For operating theatres that may have been rebalanced to achieve negative pressure environments, it should be noted that all existing settings of dampers, VSD's etc. should be locally marked and recorded to allow easier re-establishment when returned to normal usage.
5. Review of existing site asset management plan:
- To identify the associated plant and equipment to the designated surge areas to determine/create a temporary maintenance schedule to capture increased service reliability
- To also include new, temporary requirements such as
i.) Intermittent monitoring of oxygen concentration levels within each bed area via handheld device
ii.) Increased maintenance frequency to supporting infrastructure
6. LHD Capital Works teams to compile brief to outline required changes. This will likely involve Group 3 FFE and the associated timeline for completion. Infection Prevention and Control delegate with recognised expertise must form part of this group.
7.Brief submitted for approval to the facility General Manager / LHD Chief Executive
8. On approval to proceed, works may commence.
9. Execution of any/ all works as identified above may require:
- Contractor to produce records or install/test sheets for any works undertaken.
- For any services being recommissioned, LHD witnessing is to be offered for general services, Medical gas works must be witnessed as per Australian Standards.